
Patent Linkage and Article 76 Proceedings in China: A Litigator's Perspective
In Short
The Situation: The NMPA and CNIPA recently jointly issued the Implementation Measures for the Early Resolution Mechanism for Drug Patent Disputes. The CNIPA and China's Supreme People's Court have each issued, separately, rules for an Article 76 Proceeding that a patent holder could opt for against a generic drug applicant.
The Result: The three recent legal rules have unveiled detailed mechanisms of China's patent linkage system. Unlike in the United States, patent holders may choose to commence an Article 76 proceeding by means of a civil action before the court and/or an administrative determination before the CNIPA. The NMPA will grant a nine-month stay for marketing approval of the generic drug application pending a judicial or administrative decision. The first generic drug applicant that successfully challenges the validity of listed patents and obtains marketing approval will be granted a 12-month market exclusivity.
Looking Ahead: International patent holders should be mindful in selecting patents for listing, actively prepare for patent validity challenges for the listed patents, and be ready to commence an Article 76 proceeding by means of a civil action and/or an administrative determination within 45 days after a generic market application is disclosed.
China has recently unveiled its own version of a patent linkage system that has been highly watched by stakeholders and industry observers. This new system will also create a new form of pharmaceutical patent litigation in China.
Article 76 of China's Fourth Amendment of Patent Law (effective June 1, 2021) for the first time introduced a "dual-track" early resolution mechanism for resolving drug patent disputes during the marketing approval process of generic drugs, allowing innovative drug patent holders to institute a civil action or an administrative determination against generic drug applicants in order to stay the marketing approval process for generic drugs.
Details of China's early resolution mechanism were further outlined in three legal measures that were promulgated and came into effect on July 4 and 5, 2021, namely:
- Implementation Measures for the Early Resolution Mechanism for Drug Patent Disputes ("Trial for Implementation") jointly issued by the National Medical Products Administration ("NMPA") and China National Intellectual Property Administration ("CNIPA");
- Administrative Adjudication Measures for the Early Resolution Mechanism for Drug Patent Dispute issued by the CNIPA; and
- Provisions on Several Issues Concerning the Application of Law in the Trial of Civil Cases of Patent Disputes Related to Drugs Applied for Marketing Authorization issued by the Supreme People's Court of China.
Article 76 of the Chinese Patent Law has been commonly perceived as China's Hatch-Waxman Act. However, the early resolution mechanism under Article 76 and subsequent implementing measures have also featured a number of marked Chinese characteristics. International pharmaceutical patent holders and practitioners need to heed this and also take steps to manage how this new mechanism will work in the larger context of the Chinese patent litigation ecosystem. As generic drug competition intensifies in China, industry stakeholders may need to consider how to leverage this new mechanism in China and address possible accelerated invalidity challenges in China while coordinating parallel proceedings in the United States and elsewhere.
Overview of Article 76 Proceedings
As part of the new regulatory framework, the NMPA has set up the China Patent Information Registration Platform for Marketed Drugs ("CPIRPMD"), equivalent to the Orange Book in the United States, to allow a marketing authorization holder to register patent information with respect to (i) chemical drugs (compound patents of active ingredients, composition patents containing active ingredients and indication patents), (ii) biosimilars (sequence structure patents of active ingredients and indication patents), and (iii) traditional Chinese medicine (compound patents, medicine extract patents, and indication patents) within 30 days upon receipt of marketing approval of those drugs.
A generic drug applicant seeking marketing authorization will be obligated to file a patent declaration based on the status of the listed patent (if any) on the CPIRPMD, in particular:
- Type I: no listed patent on the CPIRPMD;
- Type II: listed patents that have expired, been declared invalid, or licensed to the generic applicant;
- Type III: the applicant promises not to market the generic drug prior to the expiration of the listed patents; and
- Type IV: the listed patents shall be declared invalid (Type VI-1), or the generic drug does not fall into the scope of protection of the listed patents (Type VI-2), where the applicant shall provide a chart showing the comparison of technical solutions of the generic drug being filed for application and claims of the listed patents.
As of September 13, 2021, the CPIRPMD has received registration for 527 chemical drugs, 77 biosimilars, and 239 traditional Chinese medicines, where 323 patent declarations, including 234 Type I declarations, 14 Type II declarations, 40 Type III declarations, 15 Type IV-1 declarations and 20 Type IV-2 declarations, have been filed under 279 generic drug applications.
Upon receiving generic drug applications with a Type IV declaration, a patent holder may choose to litigate whether the generic drug falls into the scope of protection of the listed patents by means of a civil action before the Beijing Intellectual Property Court ("Beijing IP Court") or an administrative determination before the CNIPA within 45 days after a Type IV declaration is published on the CRIPRMD (instead of after receiving notifications from generic drug applicants). The NMPA will provide a 9-month stay period for marketing approval of the subject generic drug application pending a judicial or administrative decision (cf. a 30-month regulatory stay in the United States). Marketing approval of the generic drug shall only be suspended if the court judgment or administrative ruling, whether received within nine months or after nine months but in the course of the marketing approval process, held that the generic drug application falls within the scope of protection of the listed patent.
It is interesting to note that only the first generic drug that successfully challenged the validity of listed patent and obtained marketing approval, i.e., filed a Type IV declaration, invalidated the relevant listed patents and obtained marketing approval, will be granted a 12-month market exclusivity period (cf. 180-day market exclusivity period in the United States). Nevertheless, such market exclusivity period shall not exceed the patent term and technical review of other generic drug applications shall continue.
In fact, Article 76 proceedings also apply to applications for biosimilars, although there will be neither a 9-month stay period for marketing approval pending a judicial or administrative decision, nor a 12-month market exclusivity period.
Choosing Between a Civil Action and an Administrative Determination
The dual-track mechanism may pose some challenges to the patent holder in deciding whether to commence an Article 76 proceeding by means of a civil action or an administrative determination. Having said that, the patent holder may weigh the pros and cons of each mechanism before deciding whether to opt for a civil action or an administrative determination.
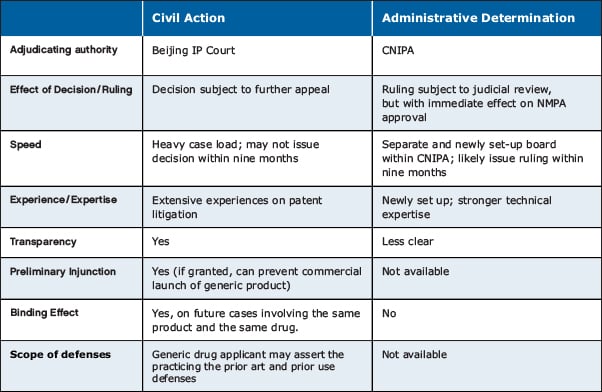
The following summarizes several salient issues a patent holder should consider in deciding whether to commence a civil action or an administrative determination:
- Declaratory in nature: As there is no concept of "artificial act of infringement" for a patent holder to establish an act of infringement under Article 76, a patent holder may only seek a declaratory judgment that a generic drug application falls within the scope of the listed patents and not damages award or injunction grant (other than preliminary injunction under civil action). On the other hand, if a patent holder and its interested parties do not commence Article 76 proceedings within 45 days after marketing approval is disclosed, a generic drug applicant may also, by means of a civil action or an administrative determination, seek a declaratory judgment that the generic drug application does not fall into the scope of protection of the listed patent.
- Litigation readiness: A patent holder needs to be well prepared before lodging a case, especially when the case is to be filed before the Beijing IP Court. An executed power of attorney, an identity certificate of the legal representative and a copy of the applicant's certificate of good standing with notarization and legalization are required for foreign patent holders. In light of the pandemic, potential patent holders should reserve adequate preparation time. Further, as generic drug applicants are required to register their email address on the CRIPRMD, service of process of court proceedings can therefore be done by email and not via Hague Service Convention for overseas parties.
- Jurisdictional challenge: Jurisdictional challenge is commonly raised in China as a tactic to slow down litigation process. As the Beijing IP Court and the CNIPA are specially designated as the adjudication authorities of Article 76 proceedings, it is less likely for jurisdictional challenge to be accepted even if raised, thereby reducing the impact of potential blockers to legal or administrative proceedings.
- Validity challenge: A generic drug applicant may commence invalidation proceedings against certain listed patents to accelerate the marketing approval process. Although there would be no stay of an Article 76 proceeding upon the filing of a validity challenge, an Article 76 proceeding can be dismissed if the Reexamination and Invalidation Department of the CNIPA invalidates the relevant patent. An invalidation challenge generally moves quickly and can be concluded in approximately five to six months' time. A patent holder should therefore be prepared to defend the validity of relevant patents.
- Discovery and evidence production: Although there is no U.S.-style discovery in China, the new mechanism seems to provide for the shift of burden of proof, which probably makes it easier for patent holders to establish their case. In particular, during the course of a civil action, a generic drug applicant will have to submit to the Beijing IP Court a copy of all technical submissions to the NMPA for determining whether the generic drug falls within the scope of patent protection. The new rules also allow CNIPA to verify relevant evidence with the NMPA during the process of administrative determination. Each party is also required to disclose relevant materials within a prescribed time.
- Preliminary injunction: A patent holder may seek a preliminary injunction before the Chinese court to prevent a generic drug applicant from using, exploiting, offering for sale or selling the relevant generic drug. The following factors will be considered: (i) the case has a high likelihood of success considering its factual basis and legal foundation; (ii) the patent holder will suffer irreparable damage if no preliminary injunction is granted; (iii) threatened harm to the patent holder outweighs threatened harm to the generic drug applicant; and (iv) it is in the public interest to grant preliminary injunction. Once a preliminary injunction is granted, generic drug applicants will likely be stopped from commercially launching the pharmaceutical products, even after marketing approval. This can be a powerful tool for patent holders.
- Malicious lawsuit: If a patent holder has knowledge or constructive knowledge that its patent should be invalid or there is no chance to proof infringement but insists to assert its patent under Article 76 proceedings, a generic drug applicant will have a cause of action to seek damages against the patent holder before the Beijing IP Court.
Two Key Takeaways
1. Under China's patent linkage system, the market exclusivity period is tied to a successful patent invalidity challenge, and it is not clear whether such invalidity challenge needs to be filed after the Type IV declaration. Therefore, patent holders need to be mindful in selecting certain patents for listing on CRIPRMD, while keeping others away from Type IV challenges. As to the patents that are selected for listing, patent holders need to be well prepared to defend their validity.
2. Legal practitioners should coordinate potential parallel proceedings in China and other jurisdictions, such as the United States, at an early stage, being mindful of differences in civil procedure or legal systems that may lead to contrary positions taken, or strategies and arguments adopted in a non-U.S. litigation. U.S. practitioners may also make use of the potential shift of burden of proof offered by the new mechanism and seek to adopt strategies that are not originally applicable due to the lack of U.S.-style discovery.




